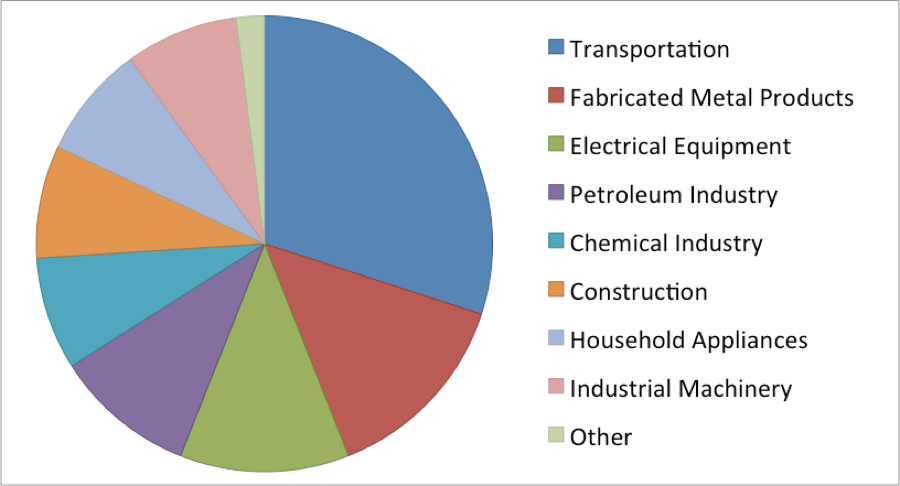
Way back in elementary school when you thought of nickel, the first image that popped into your head was probably that nice shiny 5¢ coin. Even though we hardly carry change purses anymore, nickel is still ever present in our day-to-day lives. From the shiny stainless steel trim on your
Enhanced methods for nickel recovery from low-grade ores and bleed streams
Topics: nickel, Refinery Optimization
Electrowinning is the process of ‘winning’ dissolved metals from solution by passing an electrical current through an electrolyte containing said metal. The fundamentals of the electrowinning process have been discussed in a previous blog. The relative ease of recovery depends on the electrochemical potential of the target metal relative to the Standard Hydrogen Electrode (SHE) which is defined by the following reaction:
Topics: electrowinning, nickel, emew, Refinery Optimization
Topics: electrowinning, nickel, emew, Refinery Optimization
What pops into your mind when you see the word, "liberation"?
---
The word liberation comes from the latin word, liberatus, which literally means "to set free" or "to deliver", and can easily apply to myriad of different topics from politics and religion to science and industry.
Particularly in the copper industry, the word liberation applies to the act of freeing copper specifically from refinery bleed electrolyte. The process that starts with mining and carries on through smelting and electrorefining is complex with many process operations. Let's take a closer look at the story of 'liberating copper'.
On average, mined ore contains about 1% copper and in order to recover this copper from the rock it requires several physical and chemical processing and refining steps in order to produce market ready copper metal.
Topics: copper, nickel, Refining, Refinery Optimization