Electrocoagulation (EC) is an electrolytic process that is used mostly for wastewater and industrial process water treatment. Due to its unique ability to remove contaminants such as emulsified oils, suspended solids and dissolved metals, electrocoagulation has become one of the fastest growing technologies in this area. There are various devices and systems that comprise the basic principle of electrocoagulation which I would like to explain in this article.
Let’s use as an example an industrial wastewater treatment case and assume that we have a bath filled with contaminated solution. Contaminants can be organic materials, dyes, wash water, ink, heavy metals and others. A series of anodes and cathodes are submerged into the solution and a DC voltage is applied between the electrodes.

In electrocoagulation, contrary to some other electrolytic processes such as electrowinning, the anode is made of a corrosive metal, usually iron or aluminium. When connected to the power source the anode starts to oxidize and corrode into solution as metal cations. At the same time, water at the cathode is hydrolyzed into hydrogen gas and hydroxyl groups. Metal ions complex with hydroxyl groups to form large flocs which also entrain suspended solids, emulsified oils and other contaminants. The released ions also serve to neutralize surface charges of particles and by doing so facilitate coagulation. The newly formed flocs are heavier than water and sink to the bottom of the tank. Subsequent sedimentation and filtration processes are used to separate the flocculated solids from the supernatant water to achieve the required water quality objectives.
During the process of coagulation and over time the anodes will completely dissolve and will require replacement. In some systems, the whole series of anodes-cathodes will be removed from the tank and a new set submerged.
In the electrocoagulation process, ions are the product of the electrolytic reaction therefore electrocoagulation does not require any other oxidizing or reducing agents, and does not generate any additional pollution to the environment.
This simple process is low cost, has a small footprint and offers excellent operating performance over a range of contaminants and metal concentrations.

HSJ electrocoagulation systems offer many advantages compared to other wastewater treatment technologies:
- Short installation time, low operating costs and excellent treatment performance. The main capital costs are for the equipment and a small amount of civil work on site. The operating costs are primarily power and metallic plate.
- Small footprint for equipment with small amount of civil work, considerably less than other processes.
- Highly automated: simple and user-friendly operation with remote monitoring.
- A wide range of operating conditions with resistance to shock loading; suitable for a variety of contaminants, including manganese, chromium, lead, cadmium, copper and other heavy metals regardless of hydraulic loading and water quality.
- High contaminant removal rate with only a small amount of sludge produced. The removal rate for manganese and chromium is superior to other technologies with less sludge produced.
- Simple operation and low maintenance by consuming power instead of chemical agents, with the ability to operate day in / day out with easy start / stop.
- Stable treatment performance even if water quality or quantity changes.
The basic process of wastewater treatment with HSJ electrocoagulation system would typically resemble the following:
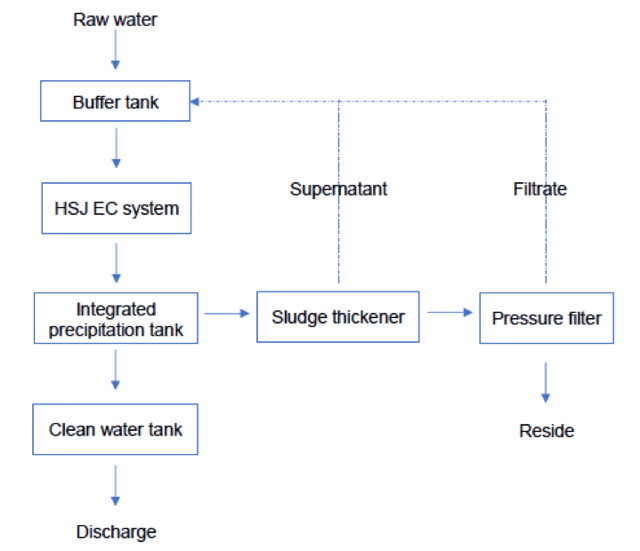
HSJ electrocoagulation technology has been applied successfully in a wide variety of industrial applications including zinc smelting, mining and beneficiation, lead smelting, copper smelting, gold refining, manganese electrolysis, electroplating and chemical production.
To learn more about electrocoagulation and industrial wastewater treatment applications please contact us or visit www.emew.com.
Sources:
https://en.wikipedia.org/wiki/Electrocoagulation








