The electro-refining of copper is a multi-step process that starts with copper concentrate that is smelted to produce impure copper anodes, which are then
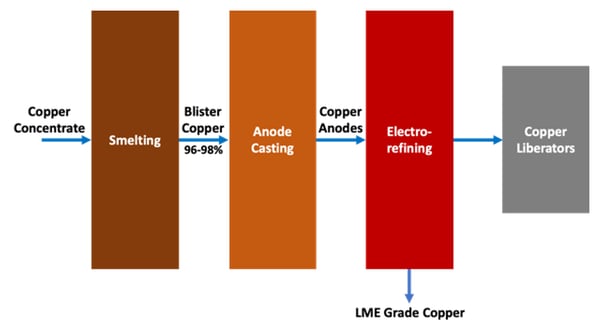
A key aspect of copper electrorefining is the control of copper concentration and impurities in the electrolyte. The dissolution of copper at the anode is inherently faster than the electroplating of copper at the cathode, hence copper tends to build up in the electrolyte and needs to be controlled. Without a proper electrolyte “bleed”, the copper would continue to build up along with impurities such as arsenic, antimony
To download fill the form below
Conventional electrowinning technology is commonly used for copper liberator cells to control the copper concentration and impurities
- They are often plagued with low current efficiency
- They produce an impure cathode, which must be recycled resulting in increased working capital, and reduced profitability.
- There are serious health and safety concerns from the acid mist and potential for arsine generation.
- The formation of copper liberator sludge which collects at the bottom of the liberator cells and must be manually removed which is labor intensive and a serious health and safety risk due to the high arsenic and antimony.
A significant advancement in copper liberator technology involves the use of
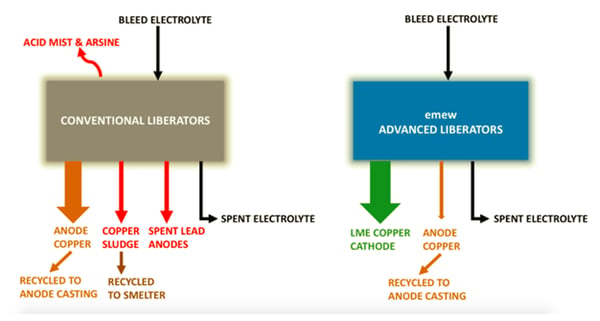
The benefits of
- Fewer recycles
- Lower working capital
- Better impurity control
- Increased profitability
- Improved health and safety
The investment to upgrade an existing
A copper refinery bleeds 80m3/day of electrolyte to conventional liberator cells. The refinery operates a multi-stage liberator circuit that depletes the copper in the electrolyte from 45 g/L down to 1-3 g/L in three stages. The first or “primary” stage of copper liberation depletes the copper from 45 g/L to 30 g/L, a ‘bite’ of 15 g/L. The “secondary” stage of copper liberation depletes the copper from 30 g/L to 15 g/L, while the third or “tertiary” stage of copper liberation depletes the copper from 15 g/L to 1-3 g/L. The refinery has undergone several upgrades and optimization projects over the years and is now able to produce saleable LME grade copper cathode from their primary liberator circuit. The copper from their secondary liberator circuit is recycled to the anode furnace, whereas the copper sludge produced from their tertiary liberator circuit can only be recycled back to the smelter. Refinery management is concerned with the health and safety of the tertiary liberator circuit due to monthly refinery evacuations from arsine alarms, extreme personal protective equipment (PPE) required by operators to clean the copper sludge from the cells, not to mention the low current efficiency and high working capital. The refinery manager contacted
The capital expenditure for an
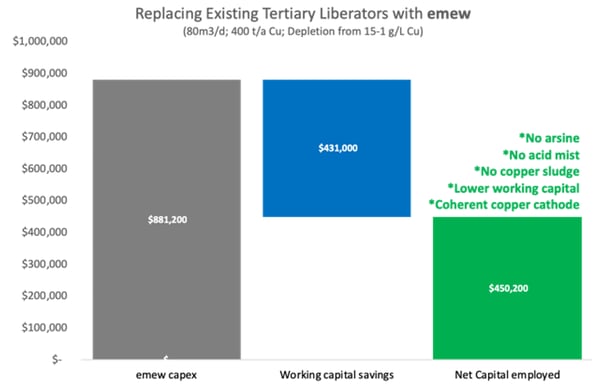
Working capital is just one impact of the recycle of the tertiary copper sludge,
Assuming current TC/RC’s of $90/tonne and $0.09/lb, this works out to USD 115,831 annually in lost revenue from TC/RC’s by recycling the tertiary copper sludge.
Furthermore, the recycle of copper to the smelter inevitably results in some smelter loss which is typically 1.5-2%. We have assumed 1.5% smelter loss at a cost of USD 37,706.
Lastly, the current efficiency in conventional tertiary liberator circuits is notoriously low. We have assumed 50% current efficiency in the tertiary liberators with conventional electrowinning, compared to 90% with
The effect of these parameters on annual operating cost can be seen in the diagram below. Overall
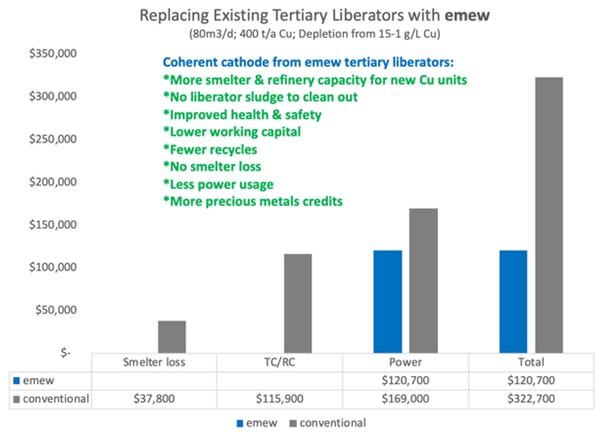
Given the savings in working capital and operating costs, the decision to upgrade tertiary liberator cells to
- emew cells are completely enclosed eliminating acid mist from the work environment
emew cells can safely deplete to low copper concentrations without the risk of deadly arsine exposureemew cells produce coherent, harvestable, saleable cathode rather than tertiary copper sludge
This case is even more compelling if the current liberators need to be upgraded or there are tougher Occupational Health requirements.





