In one of the previous posts I described the basic principle of conventional electrowinning. Today I would like to talk about the differences between conventional electrowinning and proprietary emew technology, which is based on fundamental electrowinning tenets but offers so many additional benefits.
In conventional electrowinning alternating planar anodes and cathodes are placed into a solution bath and electrolyte is slowly circulating or standing still. Positively charged ions called cations slowly make their way towards the cathode where they are reduced (i.e. gain electrons) and plate out as metal. The metal cathodes are harvested at the end of each plating cycle to recover the desired metal. Sounds good? Well, not so much. There are some problems and inefficiencies associated with this convential process.
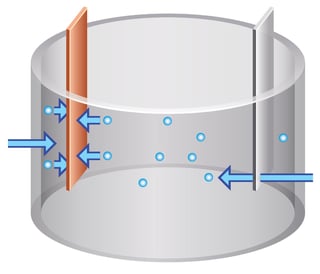
Since the solution is not circulating inside the bath, the deposition of cations happens just in the area that are very close to the cathodes. As the target metal (let's say copper for example) plates out of solution onto the cathode, zones of depletion begin to form. These so called 'depletion zones' are areas of lower concentration of the target metal (for example, copper) than in the 'bulk' solution. If there are other metal ions also present in the solution (for example, cadmium, or arsenic, or lead), these will also begin to plate onto the cathode instead of copper resulting in impurities. The metal plating process is relatively slow and as the concentration of the target metal decreases, the plating process slows down. Another thing that happens is that the relative concentrations of impuries in solution relative to the target metal begin to increase and have a greater probability of being plated onto the cathode. This process is all very well if the 'delta' or change in target metal concentration is relatively small. For example, from 40 g/L copper to say 30 g/L copper because the concentraton of copper is considerably greater, perhaps an order of magnitude or more, than any impurities. If the concentration of target metal is approximately the same or lower than any of the impurities, the cathode product will be increasingly impure. As a result, conventional electrowinning doesn't work well for solutions with low concentrations of metals and/or with high concentrations of impurities.
Let's take a look now at a more effective way to overcome these limitations using an advanced electrowinning cell design originally developed by emew over 20 years ago. The emew cell, a clever acronym for emew Electrowinning, is a cylindrical electrowinning cell in which the electrolyte solution is rapidly circulated past the anode and cathode at a very high flow rate enhancing mass transfer and allowing for improvements in efficiency and recovery. The cylindrical structure of the emew cell allows a greater selectivity and purity across a wide range of metals and even in highly contaminated waste water streams. In stark contract to conventional electrowinning cells, the emew cell is designed such that target metal cations are always presented to the cathode even at low concentrations or in the presence of high concentrations of impurities. The purity that can be achieved with an emew cell is typically 99.99% but can be as high as 99.999% or greater while in conventional electrowinning the purity depends on the initial purity of the solution, the 'delta' of depletion and the relative concentrations of impurites. For this reason, emew is the clear choice to recover high purity metals from low concentration, complex, mixed-metal streams that are often found in refineries, industrial wastewater and other waste streams.
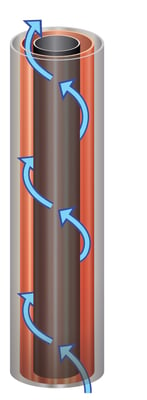

Conventional electrowinning plants are open systems. During the electrowinning process, oxygen bubbles are formed on the anode. The bubbles ultimately burst producing what is known as “acid mist”. This acid mist is extremely toxic and a known carcinogen that poses a serious health risk to employees and visitors. In addition to this, acid mist is corrosive causes ongoing damage to factory parts and results in a lot of money spent on maintenance. The emew electrowinning system on the other hand is an entirely closed system, where every emew cell is capped and isolated from the environment. This eliminates acid mist and creates a safe environment for employees and visitors alike.
On top of this emew offers real-time, remote performance monitoring of all emew plants to ensure that they operate at above design and ensure the highest safety and environmental standards are met. All emew plants are monitored 24 hours a day and notifies an operator about process adjustments to be made or emergency situations. The system provides real time information and weekly reporting mechanisms to help operators and management run an efficient, cost-effective operation.
While both electrowinning technologies are widely used, emew electrowinning has some clear advantages over conventional electrowinning. emew offers higher purity, improved recovery, increased efficiency and lower operating costs over conventional electrowinning. emew plants are easily customizable to suit a wide array of requirements and production rate and are easily expandable. Last but certainly not least, emew offers uncompromised safety and environmental performance for advanced metal recovery operations.
To download fill the form below






