Why is it easy to electrowin some metals, and almost impossible to electrowin others?
In a few minutes, I will explain why. The answer lies in the Electrochemical Series, and its relation to electrowinning.
Electrowinning is the process of removing metals from solution by electrolysis.
In even simpler terms, it is the process of giving an electron to a positively charged metal ion, or cation, in solution. The act of gaining electrons, until net zero charge, is how the metal cations are electrolytically plated as a solid metal, out of solution.
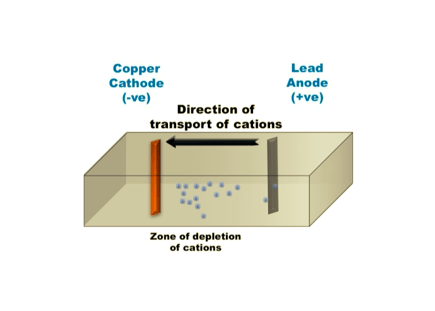
Different metals lose or accept electrons at differing rates.
In general, the more willing a metal is to accept electrons, the easier it will be electrowon from a solution.
The electrochemical series is a list of different elements and their standard reduction potentials, which tells you how easily these elements gain electrons.
The significance of putting the elements in order of their standard reduction potentials is that you can see what metals are most likely to electrowin when in solution.
On the series shown here, zinc is the last metal up the series that can be electrowon from solution; anything above zinc cannot.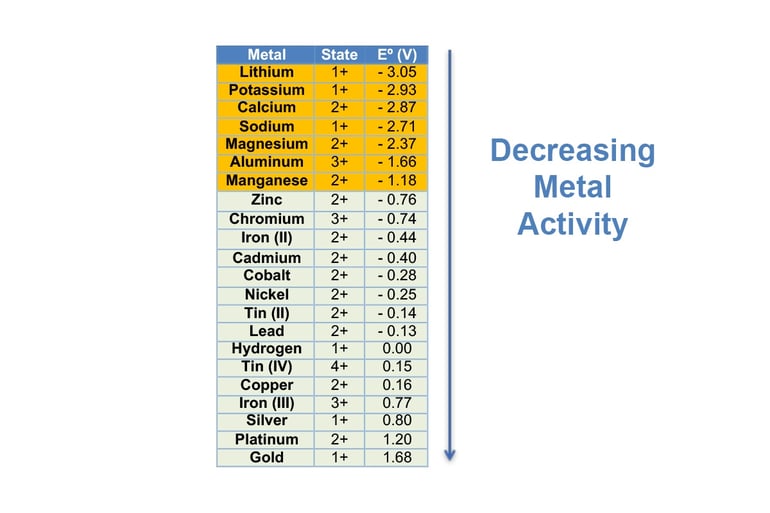 Let’s say you have a solution with equal concentrations of Silver and Copper.
Let’s say you have a solution with equal concentrations of Silver and Copper.
 Let’s say you have a solution with equal concentrations of Silver and Copper.
Let’s say you have a solution with equal concentrations of Silver and Copper.Because Silver is lower on the series, it means it will gain an electron more easily, becoming Silver metal and plating out of solution.
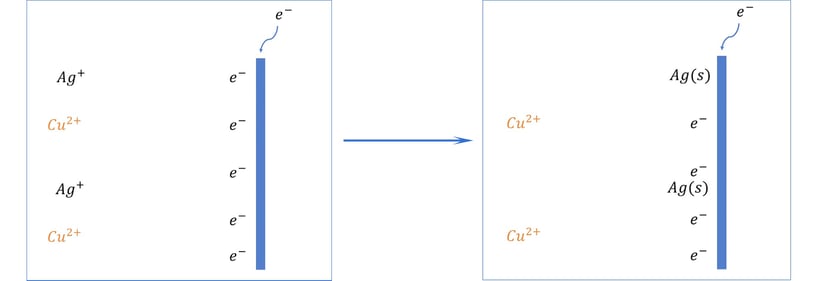
Once all the silver is depleted, then the copper will start plating, if there is nothing else in solution with a more positive reduction potential.
When determining the feasibility of electrowinning a specific metal from solution, it is important to consider the other impurities in solution, and their reduction potential relative to the target metal. As we discussed, metals with the most positive reduction potentials will electrowin most easily, which means they will plate out from the solution before metals with less positive reduction potentials.
FREE Download - emew Liberators for Copper Refineries
To download fill the form below
There are two other important things to highlight on the electrochemical series. You will notice that there are two Iron species.
Iron can be present in aqueous solution as Ferrous, that is Iron (II), or Ferric, that is Iron (III).
The Ferrous/Ferric redox couple is an important consideration for electrowinning since Iron (III) is easily reduced to Iron (II) at the cathode, but Iron(II) will oxidize back to Iron (III) at the anode.
This is a ‘do nothing’ reaction that only consumes electricity which is why it is important to minimize the amount of ferric in solution prior to electrowinning.
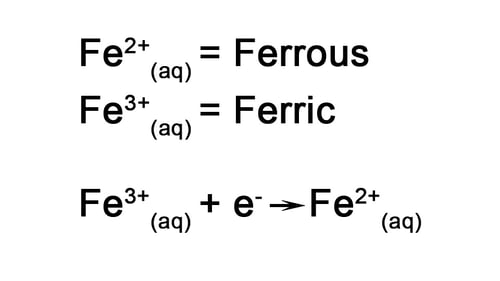
The final point to make is that the closer elements are to each other on the electrochemical series, the more likely they are to plate together, all else being equal.”
Hopefully, this helps you understand the important role the Electrochemical series plays in predicting the ability of a metal to be electrowon.
If you want to learn more about electrowinning please check out our other blog about electrowinning - Electrowinning 101.





