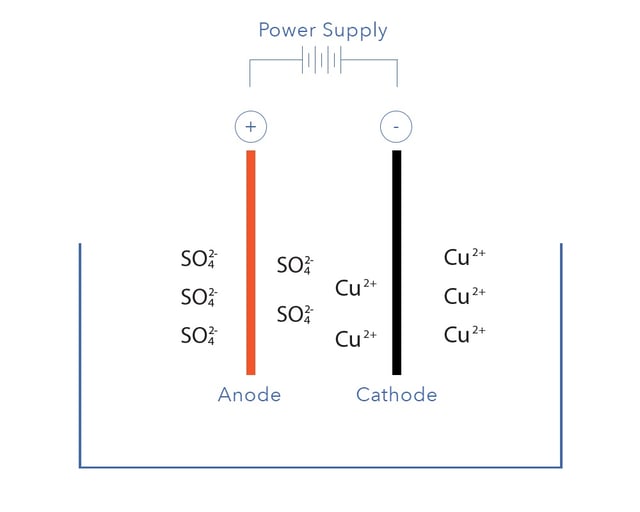
Electrowinning is known as an electrolytic process because it involves electrodes submerged into an electrolyte.An electrolyte is simply a conductive solution formed by dissolving positively and negatively charged ions.
For example,
copper and
sulphate. When a voltage is applied to the solution the positively charged cations (like copper) move towards the cathode, while the anions move towards the anode. The relative tendency of a metal to plate depends on its position in the electrochemical series.
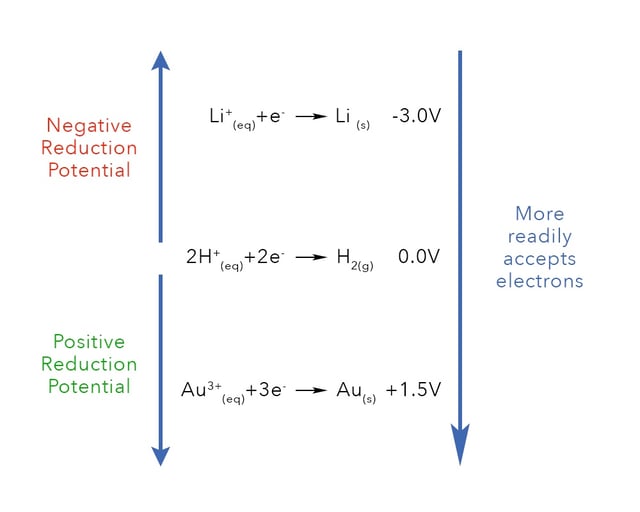
The electrochemical series lists a particular metals ability to accept compared to standard hydrogen electrode which is defined as 0.0 V. Metals that are more willing to accept electrons have a positive reduction potential are more easily plated and considered more nobel. Metals that are more willing to lose electrons have negative reduction potentials, are less easily plated and considered more reactive. Metals like gold, silver and copper for example have positive reduction potentials and are recovered quite easily by electrowinning, whereas metals like lithium and zinc have negative reduction potentials and are not as easy to recover.
In conventional electrowinning, alternating planar anodes and cathodes are placed into a static or slow moving electrolyte bath. Let’s assume an electrolyte of copper sulphate ions.
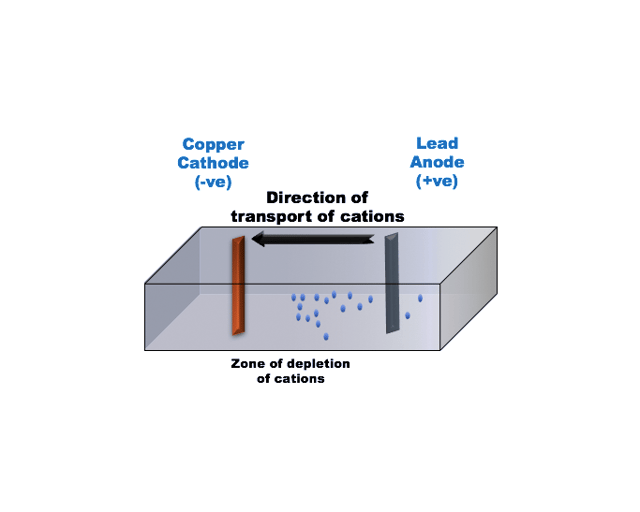
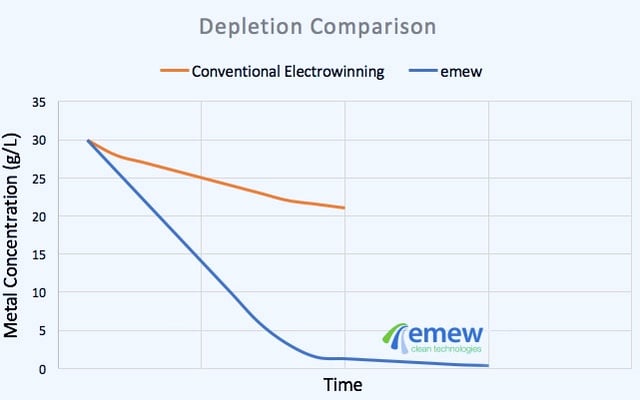
With a voltage applied, copper cations migrate towards the cathode where they are plated as pure copper metal. As metal builds-up on the cathode the concentration of metal in solution decreases and the plating slows down. After a certain period of time, the pure copper metal is harvested from the cathodes. Unfortunately as the concentration of dissolved metal decreases, depletion zones appear next to the cathode surface. In that case, impurities that happen to be present in the depletion zone may begin to plate resulting in an impure cathode. The higher the concentration of impurities and the more positive their reduction potential, the more likely they are to plate onto the cathode.

For over 150 years, there was little change to the process of electrowinning. However in the early 90’s, Electrometals (now emewCorporation) reinvented electrowinning with the emew cell. EMEW is actually an acryonym that stands for ElectroMetals ElectroWinning. Now it has become a well-established brand and the name of our company.

The emew cell is an advanced electrowinning technology that uses cylindrical cells with high flow rate to enhance the rate of mass transfer, overcoming issues associated with depletion zones and enabling production of high purity metals even in presence of impurities. Pushing the limits even further, emew continues to develop the technology including high surface area cathodes that enable recovery of metals down to the part-per-million level. emew cells have expanded the applicability of electrowinning beyond just metal refining to recycling, waste and wastewater treatment and even into hi-tech industries like semiconductors.
That’s all for now. We’d love to hear your feedback so feel free to leave a comment.













